Streamline Your FDA Compliance Journey
Explore our expert regulatory consulting services tailored for medical device success.
Regulatory Strategy Development
Crafting clear pathways to meet FDA requirements efficiently.

Submission Management
Handling your FDA filings with precision to accelerate approvals.
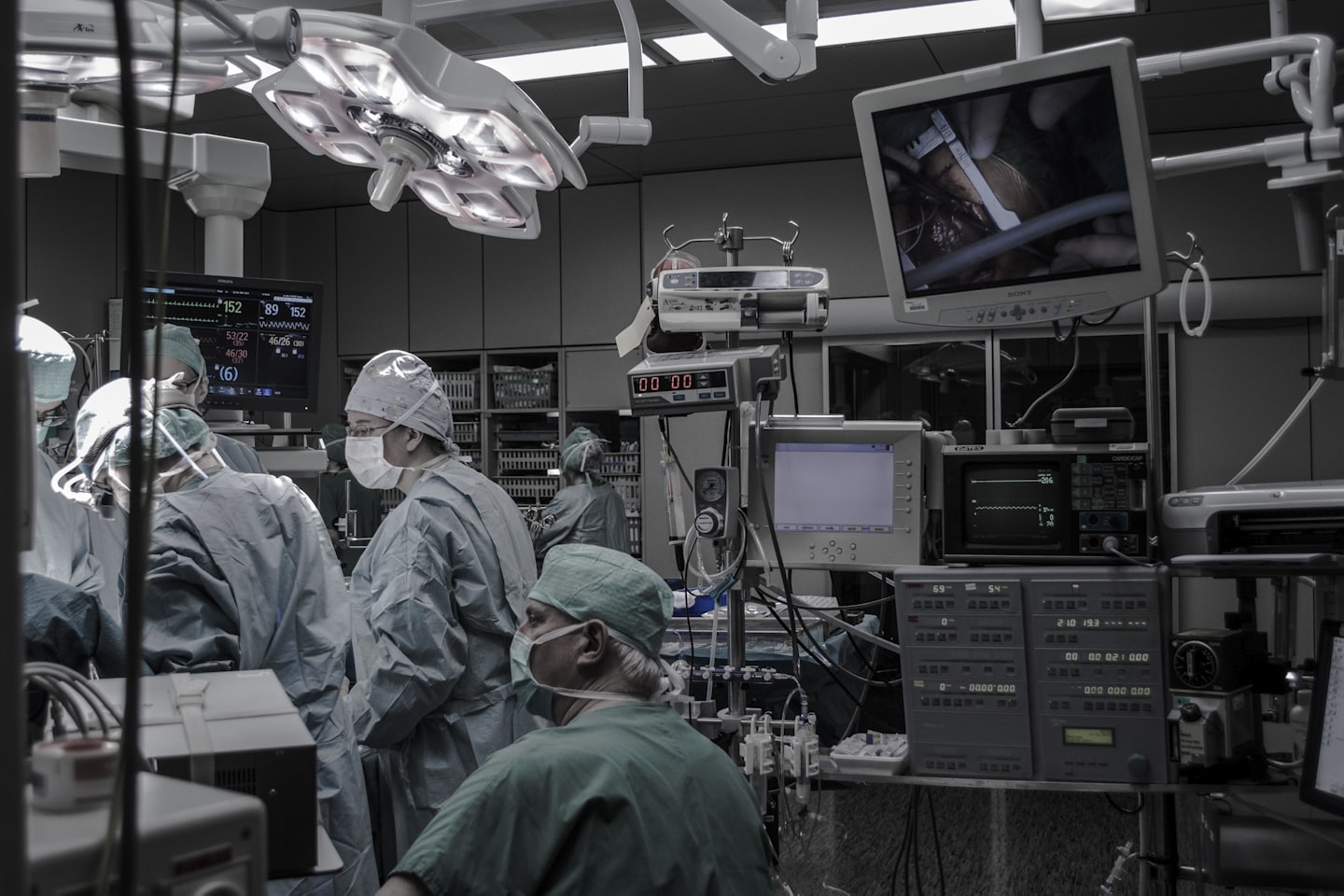
Mock FDA Audits
Preparing your team through realistic inspections to ensure readiness.
Expert FDA Inspection Preparation Services
Discover how BraunSolutions Regulatory Group leverages over 30 years of regulatory expertise to guide medical device companies through FDA compliance, submissions, and audit readiness, ensuring seamless product approvals.
Comprehensive Compliance Management
Strategically navigate FDA regulations with personalized compliance plans tailored to your medical device needs.
Mock FDA Audit Simulations
Experience realistic audit rehearsals that identify gaps and boost your team’s preparedness for real inspections.
Regulatory Support
Expert guidance for start-up medical device companies and market entry by assisting in the creation of your 21 CFR820 / ISO 13485 QMSR.
Expert Regulatory Support, Proven Results
Discover our certifications, FDA compliance expertise, and industry accolades that build confidence in our services.
FDA Submissions and More
Comprehensive submission assistance for Traditional, Special, and Abbreviated 510(k)’s, De Novo, PMA;s while ensuring adherence to medical device standards.
We also provide response assistance to 483 Observations and Warning Letter’s.
ISO 13485 Compliance
Our processes meet ISO 13485 standards, demonstrating commitment to quality management in medical devices.
Experienced Regulatory Professionals
Over 30 years of industry experience guiding successful product approvals and audit preparations.
